Aerobic flow oxidation of alcohols in water catalyzed by platinum nanoparticles dispersed in an amphiphilic polymer†
Abstract
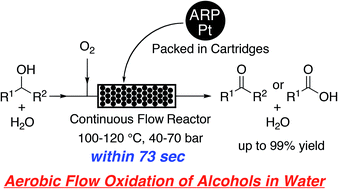
We have developed a technique for the aqueous aerobic flow oxidation of alcohols in a continuous-flow reactor containing platinum nanoparticles dispersed on an amphiphilic polystyrene–poly(ethylene glycol) resin (ARP-Pt). Various primary and secondary alcohols including aliphatic, aromatic and heteroaromatic alcohols were efficiently oxidized within 73 seconds in a flowing aqueous system at 100–120 °C under 40–70 bar of the system pressure to give the corresponding carboxylic acids and ketones, respectively, in up to 99% yield. Benzaldehydes could be also prepared selectively from benzyl alcohols by conducting the flow oxidation under the standard conditions in the presence of triethylamine. Moreover, a practical gram-scale synthesis of surfactants was realized in the aqueous aerobic continuous flow oxidation for 36–116 hours. This aerobic flow oxidation system provides a safe, clean, green, rapid and efficient practical method for oxidizing alcohols.

 Please wait while we load your content...
Please wait while we load your content...