Hydrophobic mesoporous silica-supported heteropolyacid induced by ionic liquid as a high efficiency catalyst for the oxidative desulfurization of fuel†
Abstract
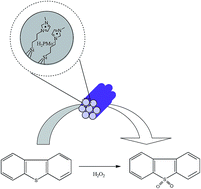
Hydrogen peroxide is usually used as an oxidant in oxidative desulfurization. However, in order to reach a high level of sulfur removal, excess of hydrogen peroxide needs to be added into the oxidative system. In this study, phosphomolybdic acid immobilized on imidazole-based ionic liquid modified mesoporous SBA-15 (HPMo-IL/SBA-15) was prepared. The as-prepared material combines the advantages of H3PMo12O40 (HPMo) and SBA-15. The high special surface area of SBA-15 could well disperse the HPMo active species. The results show that HPMo could be steadily immobilized across the surface of SBA-15 by using an imidazole-based ionic liquid. At the same time, after the introduction of the imidazole-based ionic liquid, HPMo-IL/SBA-15 material exhibited hydrophobic property. Experiments demonstrated that the hydrophobic catalyst exhibited a high activity in oxidative desulfurization. When hydrogen peroxide with a stoichiometric ratio (H2O2/sulfur mole ratio is 2) was used, the removal of dibenzothiophene (DBT) could achieve levels of above 90% at 60 °C in 90 min. When the concentration of hydrogen peroxide was slightly increased (H2O2/sulfur mole ratio increased from 2 to 2.5), the sulfur removal reached 100% in 40 min. Moreover, the synthesis catalyst could adsorb dibenzothiophene sulfone (DBTO2), which is the oxidant product of DBT. In this regard, HPMo-IL/SBA-15 served as not only a high-efficiency catalyst but also an adsorbent.
- This article is part of the themed collection: Ionic Liquids: Editors collection for RSC Advances

 Please wait while we load your content...
Please wait while we load your content...