Fluorescence resonance energy transfer between ZnO/MgO/carboxymethyl-β-cyclodextrin and Nile Red in HeLa cells – biosensing applications†
Abstract
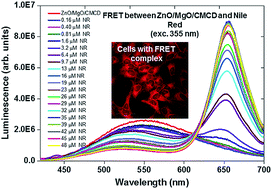
Early diagnosis remains an important problem of cancer treatment strategies. There is therefore a great need for cancer detection tests that are fast, inexpensive and do not require sophisticated laboratory equipment. For this purpose, we synthesized the ZnO/MgO core/shell nanoparticles which are relatively nontoxic, inexpensive and simple to create. We constructed biosensors based on ZnO/MgO nanoparticles which employ the fluorescence resonance energy transfer from ZnO/MgO nanoparticles (donor) to Nile Red (acceptor). Characteristic features of Nile Red luminescence are its solvatochromic and thermochromic properties. In the physiologically relevant temperature range (20–45 °C), the shift of Nile Red luminescence in the ZnO/MgO/CMCD/Nile Red complex is linear with temperature. In our experiment, thermochromic shift was 5.7 ± 1.5 cm−1 K−1. Nile Red thermochromism observed in the complex will allow us to construct a sensor capable of detecting exothermic changes and local environmental differences between normal and pathological cells. Subsequently, we studied ZnO/MgO/CMCD/Nile Red complex in vivo in biological samples. We present here, for the first time that the donor–acceptor energy transfer is affected by the intracellular or extracellular locations of the nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...