A novel three-dimensional gold catalyst prepared by simple pulse electrodeposition and its high electrochemical performance for hydrogen peroxide reduction
Abstract
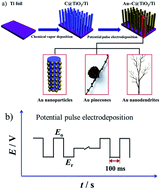
Novel Au nanoparticles (NP), Au pinecones (PC) and Au nanodendrites (ND) supported on carbon coated titanium dioxide (C@TiO2) nanoarrays were successfully obtained through a facile chemical vapor deposition of three-dimensional (3D) C@TiO2 substrate, followed by potential pulse electrodeposition of Au electrocatalysts. The morphology and structure of the open 3D Au–C@TiO2 electrodes was characterized by scanning electron microscopy and X-ray diffractometry. The different morphology of electrodeposited Au can be easily controlled by the applied potential (Eo). Electrochemical methods, including cyclic voltammetry, linear sweep voltammetry and chronoamperometry, were used to examine the catalytic activity of the electrode for H2O2 electroreduction in H2SO4 solution. The Au ND–C@TiO2 electrode exhibited the largest effective specific surface area among the Au–C@TiO2 electrodes, owing to its open nanodendritic structure allowing the full utilization of Au surface active sites. A nearly constant reduction current density of 0.655 A cm−2 was successfully achieved on the Au ND–C@TiO2 electrode at the potential of 0 V in 2.0 mol L−1 H2O2 + 2.0 mol L−1 H2SO4 solution, which was significantly higher than the catalytic activity of H2O2 electroreduction achieved previously with precious metals as catalysts.

 Please wait while we load your content...
Please wait while we load your content...