Casein glycomacropeptide hydrolysate exerts cytoprotection against H2O2-induced oxidative stress in RAW 264.7 macrophages via ROS-dependent heme oxygenase-1 expression
Abstract
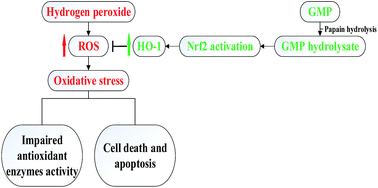
The aim of this study was to investigate the antioxidant potential of bovine casein glycomacropeptide (GMP) and its hydrolysate, as well as to determine effects of GMP and its hydrolysate on hydrogen peroxide (H2O2)-induced oxidative stress in RAW 264.7 macrophages. In comparison with native GMP, GMP hydrolysate obtained with papain for 1 h hydrolysis (GHP) exerted higher free radical-scavenging capacity, ferrous ion (Fe2+)-chelating ability, and ferric reducing (FRAP) activity. GHP significantly blocked H2O2-induced intracellular reactive oxygen species (ROS) generation as well as cell death, and the cytoprotective effects of GHP were partially reversed by co-treatment with zinc(II)-protoporphyrin IX (ZnPPIX), a specific inhibitor of HO-1. GHP induced nuclear translocation of the nuclear factor-erythroid-2-related factor 2 (Nrf2) and heme oxygenase-1 (HO-1) expression in RAW 264.7 macrophages. The chemical antioxidant, N-acetyl cysteine (NAC), significantly reduced GHP-induced HO-1 expression and Nrf2 activation by blocking intracellular ROS production. Additionally, pretreatment with GHP enhanced cellular antioxidant enzymes activities of superoxide dismutase (SOD), glutathione peroxidase (GSH-Px) and catalase (CAT) in H2O2-damaged cells. The antioxidant activity of GHP may be attributed to its amino acid profiles. Compared with native GMP, GHP had higher contents of alanine, glycine, glutamic acid, aspartic acid and branched chain amino acids (BCAAs, leucine, isoleucine and valine). BCAAs-enriched GHP may possess a potential to ameliorate oxidative stress-related diseases.

 Please wait while we load your content...
Please wait while we load your content...