Cu- or Fe-catalyzed C–H/C–C bond nitrogenation reactions for the direct synthesis of N-containing compounds
Abstract
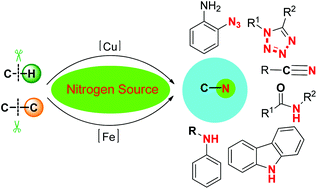
Nitrogen-containing compounds are widely present in both natural products and synthetic compounds, for example, they show up within functional materials, top-selling drugs, as well as bioactive molecules. Thus, organic chemists have paid considerable attention in developing novel methodologies for their preparation. To synthesize these compounds in a green and sustainable way, researchers have focused on the direct functionalization of hydrocarbons via C–H and/or C–C bond cleavage. Although significant progress has been made in the direct functionalization of simple hydrocarbons, direct incorporation of N-atoms into simple substrates via C–H and/or C–C bond cleavage remains challenging due to the inert chemical bonds and the unstable character of some N-sources under oxidative conditions. Azide reagents are frequently used as nitrogen source in incorporating nitrogen into carbon skeletons. Although the exact reaction pathway remains unclear, detailed mechanistic studies revealed that the carbon cation containing the azido group may exist as the key intermediate which would undergo Schmidt-type rearrangement to afford nitriles, tetrazoles, arylamines, or other kinds of nitrogen-containing compounds. Considering the high cost and toxicity of heavy metals, copper and iron, as inexpensive, readily accessible metals have already shown their unique utilities. This account attempts to focus on C–H/C–C bond nitrogenation reactions via Cu and Fe catalysis, as well as their applications in synthetic chemistry.
- This article is part of the themed collection: HOT articles in Organic Chemistry Frontiers in 2015

 Please wait while we load your content...
Please wait while we load your content...