DFT and TD-DFT studies of electronic structures and one-electron excitation states of a cyanide-bridged molecular square complex†
Abstract
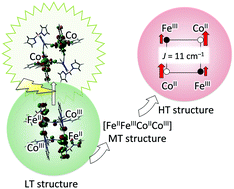
The electronic structures of a cyanide bridged Fe–Co molecular square, [Co2Fe2(CN)6(tp*)2(dtbbpy)4](PF6)2·2MeOH (1) (tp* = hydrotris (3,5-dimethylpyrazol-1-yl) borate, dtbbpy = 4,4′-di-tert-butyl-2,2′-bipyridine), which exhibits thermal and photo-induced two-step charge-transfer induced spin transitions (CTIST), are investigated in detail by density functional theory (DFT) and time-dependent (TD) DFT calculations. For the three phases observed in the experiment, three different model structures are constructed based on the geometries of X-ray crystallography analysis measured at low (100 K), middle (298 K) and high (330 K) temperatures. The calculated results elucidate that the ground states at the low and the high temperatures are diamagnetic [(FeIILS)2(CoIIILS)2] and ferromagnetic [(FeIIILS)2(CoIIHS)2], respectively. On the other hand, the one-electron transferred [FeIILSFeIIILSCoIIHSCoIIILS] state becomes the ground state at the intermediate temperature phase. A magnetic interaction between FeIII and CoII in the [(FeIIILS)2(CoIIHS)2] state is ferromagnetic and the most stable spin-coupling state is the all-ferromagnetic state. The TD-DFT calculation shows the two significant peaks of FeII t2g → CoIII eg around 800 nm. The results support that the experimental broad absorption peak at 770 nm is an inter-valence charge transfer (IVCT) band.
- This article is part of the themed collection: Molecular Magnetism themed collection

 Please wait while we load your content...
Please wait while we load your content...