Structural and magnetic tuning from a field-induced single-ion magnet to a single-chain magnet by anions†
Abstract
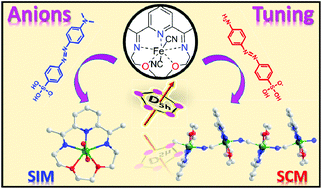
We herein report the syntheses, crystal structures, and magnetic properties of two complexes based on the anisotropic pentagonal bipyramidal FeII starting material [Fe(LN3O2)]2+, namely [Fe(LN3O2)(H2O)2][MQ]2·H2O (1) and [Fe(LN3O2)(CN)][ABSA]·3H2O (2) (LN3O2 = 2,13-dimethyl-6,9-dioxa-3,12,18-triazabicyclo[12.3.1]octadeca-1(18),2,12,14,16-pentaene; MQ− = methyl orange anion; ABSA− = 4-aminoazobenzene-4′-sulfonic anion). Compound 1 is a mononuclear material where the [Fe(LN3O2)(H2O)2]2+ cations form a one-dimensional (1D) chain by the hydrogen bonds between the bulky MQ− anions and the coordinated water molecules. With a slightly different anion, ABSA−, a cyano-bridged FeII chain is formed for compound 2. This chain has a similar structure as that in our recently reported compound [Fe(LN5)(CN)][BF4] (Chem. Commun., 2015, 51, 4360). However, compared with the reported result where the chains interact with each other through the π⋯π interactions, the chains in 2 are well isolated by the bulky ABSA− anions with the shortest interchain Fe⋯Fe distances around 12.0 Å. Magnetic investigation on 1 reveals the easy-axis magnetic anisotropy of the mononuclear FeII centre (zero-field splitting parameter D = −3.7 cm−1), which leads to the field-induced slow magnetic relaxation. For compound 2, the Fe2+ spins are antiferromagnetically coupled through the cyano bridges with a coupling constant of J = −4.13(2) cm−1 with the Hamiltonian H = −J∑Si·Si+1. AC magnetic measurements revealed the pure single-chain magnetic (SCM) behaviour of these isolated chains with an effective energy barrier of 26.1(5) K. This system represents a good example showing that the structures and magnetic properties, such as field-induced single-ion magnets, SCMs, and SCM-based magnets, can be selectively prepared by anion modification.
- This article is part of the themed collection: Molecular Magnetism themed collection

 Please wait while we load your content...
Please wait while we load your content...