“Clickable” thiol-functionalized nanoporous polymers: from their synthesis to further adsorption of gold nanoparticles and subsequent use as efficient catalytic supports†
Abstract
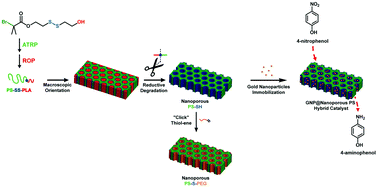
A straightforward and versatile approach towards thiol-functionalized nanoporous polystyrene frameworks is reported through the selective cleavage of a disulfide bridge at the junction between both blocks of newly synthesized polystyrene-block-poly(D,L-lactide) (PS-b-PLA) diblock copolymer precursors. This methodology requires the synthesis of a disulfide-bearing heterodifunctional initiator that allows for the production of well-defined diblock copolymers by combining atom transfer radical polymerization (ATRP) of styrene and ring-opening polymerization (ROP) of D,L-lactide. After macroscopic orientation of the copolymer precursors through channel die processing and subsequent quantitative degradation of the disulfide bridge via triphenylphosphine-mediated reduction, thiol-functionalized porous polymers are obtained. Further, “click” thiol–ene-mediated functionalization of thiol-coated pore walls within nanoporous frameworks is implemented. More interestingly, adsorption of in situ generated gold nanoparticles and subsequent and unprecedented supported catalytic reduction of a model nitroaromatic compound, i.e. para-nitrophenol, are successfully achieved. Reusability of the hybrid catalyst is also proved over a 5 run-cycle with conversion of nearly 70% within only two hours.

 Please wait while we load your content...
Please wait while we load your content...