Amphiphilic poly(disulfide) micelles and a remarkable impact of the core hydrophobicity on redox responsive disassembly†
Abstract
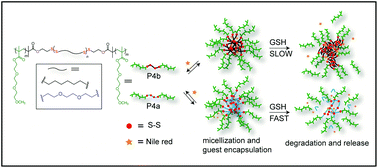
Redox-responsive amphiphilic triblock copolymers based on poly(triethylene glycol monomethyl ether)methacrylate-b-poly(disulfide)-b-poly(triethylene glycol monomethyl ether)methacrylate (PTEGMA-b-PDS-b-PTEGMA) with different hydrophobicities of the PDS block were synthesized by step-growth followed by chain-growth polymerization. By utilizing thiol–disulfide exchange reaction, polycondensation between di-thiols of varying hydrophobicities and dipyridyl disulfide produced linear telechelic poly(disulfide)s with a controlled degree of polymerization and defined functional groups at the chain-ends, which were subsequently functionalized to produce bifunctional macro-initiators to initiate atom transfer radical polymerization (ATRP) producing ABA-type amphiphilic triblock copolymers. In aqueous medium, both the polymers form micellar aggregates with an average diameter of ∼15 nm. Control experiments indicate a relatively low critical aggregation concentration (CAC) and high dye encapsulation efficiency of the less polar micellar core, reflecting the difference in hydrophobicity depending on the structure of the poly(disulfide) block. To check the impact of such structural changes on micellar disassembly and drug release kinetics, Nile Red (NR) was encapsulated as a hydrophobic probe in the interior of the micelles and glutathione (GSH)-induced disintegration of the backbone and consequently the release of the entrapped guest molecules were monitored. For micellar assembly with a relatively more hydrophobic interior, slower disintegration of the backbone and dye-release kinetics were observed compared to the micelles with less hydrophobic character, owing to slow diffusion of the polar GSH toward the more hydrophobic micellar core.

 Please wait while we load your content...
Please wait while we load your content...