Aqueous asymmetric aldol reactions in polymersome membranes†
Abstract
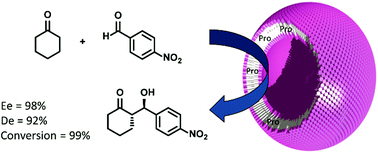
L-Proline catalysts have been immobilised in the hydrophobic domain of a polymersome via a copper(I)-catalysed azide–alkyne cycloaddition (CuAAC) reaction. Utilisation of these nanoreactors in the asymmetric aldol reaction of cyclohexanone with 4-nitrobenzaldehyde afforded the corresponding β-hydroxyketones in quantitative yields and with excellent enantio- and diastereoselectivities. The polymersomes were recycled up to five times without any loss in activity or selectivity.

 Please wait while we load your content...
Please wait while we load your content...