A new role of curcumin: as a multicolor photoinitiator for polymer fabrication under household UV to red LED bulbs†‡
Abstract
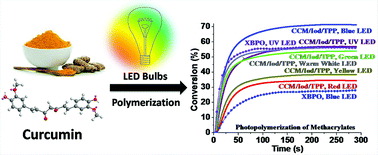
Curcumin exhibits broad ground state light absorption and can act as a photoinitiator for the free radical photopolymerization of methacrylates under air upon exposure to different household LED bulbs. The effects of temperature and various additives on the photoinitiation efficiency of curcumin-based systems have been investigated. The curcumin-based system exhibits the highest photoinitiation efficiency at 25 °C. Additives also play an important role in the photoinitiation efficiency, and well-designed systems can even demonstrate higher efficiency than the commercial type I photoinitiator [phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide (XBPO)] and type II photoinitiator [camphorquinone (CQ)]. Interestingly, the curcumin/diphenyliodonium hexafluorophosphate/triphenylphosphine combination is a capable multicolor photoinitiating system able to initiate free radical photopolymerization under air upon exposure to UV, blue, green, yellow, red, and warm white household LED bulbs. In addition, reversible addition–fragmentation chain transfer (RAFT) photopolymerization of N-isopropylacrylamide can also be achieved using a curcumin-based system under the irradiation of a blue LED bulb. The photochemical mechanisms associated with the generation of radicals from the investigated photoinitiating systems are investigated by different techniques (fluorescence, steady state photolysis, and electron spin resonance spin-trapping methods) and discussed in detail. More interestingly, the polymer sample produced through the photopolymerization process using the curcumin-based photoinitiating system demonstrates almost no toxicity to human fibroblast Hs-27 cells, endowing this photoinitiating system with great potential for the fabrication of biocompatible polymeric materials.

 Please wait while we load your content...
Please wait while we load your content...