B(C6F5)3-catalyzed group transfer polymerization of alkyl methacrylates with dimethylphenylsilane through in situ formation of silyl ketene acetal by B(C6F5)3-catalyzed 1,4-hydrosilylation of methacrylate monomer†
Abstract
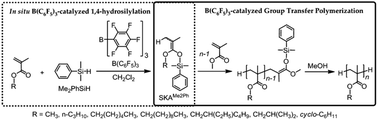
The group transfer polymerization (GTP) of alkyl methacrylates has been studied using hydrosilane and tris(pentafluorophenyl)borane (B(C6F5)3) as the new initiation system. For the B(C6F5)3-catalyzed polymerization of methyl methacrylate (MMA) using triethylsilane, tri-n-butylsilane (nBu3SiH), dimethylphenylsilane (Me2PhSiH), triphenylsilane, and triisopropylsilane, nBu3SiH and Me2PhSiH were suitable for producing well-defined polymers with predicted molar masses and a low polydispersity. The livingness of the GTP of MMA using Me2PhSiH/B(C6F5)3 was verified by the matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) measurement of the resulting polymers, kinetic analyses, and chain extension experiments. The B(C6F5)3-catalyzed GTP using Me2PhSiH was also applicable for other alkyl methacrylates, such as the n-propyl, n-hexyl, n-decyl, 2-ethylhexyl, iso-butyl, and cyclohexyl methacrylates. The in situ formation of the silyl ketene acetal by the 1,4-hydrosilylation of MMA was proved by the MALDI-TOF MS and 2H NMR measurements of the polymers obtained from the B(C6F5)3-catalyzed GTPs of MMA with Me2PhSiH or Me2PhSiD, which was terminated using CH3OH or CD3OD.

 Please wait while we load your content...
Please wait while we load your content...