Synthesis, postmodification and fluorescence properties of reduction-cleavable core-couplable miktoarm stars with a branched core†
Abstract
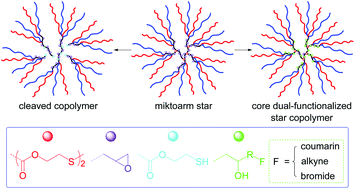
Novel amphiphilic (PEG)m(PCL)n (m ≈ n ≈ 23) miktoarm stars with a disulfide-linked epoxy-functionalized branched core were controllably synthesized via three step reactions comprising self-condensing vinyl polymerization via reversible addition–fragmentation chain transfer, epoxy-carboxyl coupling reaction and ring-opening polymerization. The star copolymers were characterized by 1H NMR spectra, GPC-MALLS, DSC and reduction-triggered degradation. Postpolymerization modification via epoxy-carboxyl/phenol coupling reactions allowed attaching dual-reactive functionalities including coumarin, alkyne and alkyl bromide onto the branched core. In water and THF–water mixtures, a coumarin-modified star copolymer could aggregate into some intriguing morphologies including hyperbranched micelles and large vesicles due to the influence of solvent polarity on aggregation behaviors. Owing to the differences in the isolation of fluorophores from solvents and restricted molecular motion, coumarin-functionalized stars exhibited adjustable fluorescence properties in water and THF–water mixtures, and their aqueous solution had a maximum quantum yield (ΦF = 44.2%). The solutions of star copolymers and their reduction-cleaved copolymers were of different hydrodynamic diameters, and the ΦF(star)/ΦF(cleaved copolymer) values were 1.85 (in THF), 3.00 (in water) and ranged between 1.29 and 2.58 in THF–water mixtures, revealing that the aggregation behaviors and fluorescence properties were strongly dependent on polymeric architecture, location of fluorophores and solvent polarity. Our study affords a versatile method to construct functional miktoarm stars with a multi-reactive branched core, and coumarin-functionalized star copolymers may have a great potential as solvent polarity and reduction dual-sensitive imaging materials in “green” ink, coatings and nanocarriers for biomedical applications.

 Please wait while we load your content...
Please wait while we load your content...