A 3-D multicellular tumor spheroid on ultrathin matrix coated single cancer cells provides a tumor microenvironment model to study epithelial-to-mesenchymal transitions
Abstract
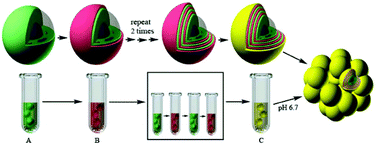
We report a 3D tumor spheroid model to study epithelial-to-mesenchymal transition (EMT) using ultra-thin matrix coated single cancer cells. The spheroid was characterized by light microscopy, fluorescence imaging, SEM, TEM and zeta potential. We then studied EMT related cellular CD47, mTOR, ERK, and HIF signaling. We compared (1) cells inside a multicellular tumor spheroid (3Dis), (2) cells migrated from the multicellular tumor spheroid (3Dms), and (3) 2D monolayer-cultured cells (2Ds) in EMT. 3Dis exhibited an abnormal nucleus with the occurrence of cell damage and accumulation of lipid droplets, and higher expression of the integrin-associated protein and dissociation of the mTOR complex than those of 2Ds and 3Dms. We examined the expressions of E- and N-cadherin, two EMT related factors, and found over-expression of E-cadherin in 2Ds, high N-cadherin in 3Dis, and high E- and weak N-cadherin in 3Dms. This implied that our model could trigger EMT. By injecting cells into a xenograft model, we found transitions of 2Ds from N-cadherin negative to positive and transitions of 3Dis from positive to negative, but not in 3Dms. 2D cells exhibited higher progression than other cells. This suggests that the in vivo 3D tumorigenesis environment could limit the capacity of cancer metastases for 3Dis but trigger the EMT process quickly for 2Ds and 3Dms.

 Please wait while we load your content...
Please wait while we load your content...