The effect of N-methylation of amino acids (Ac-X-OMe) on solubility and conformation: a DFT study†
Abstract
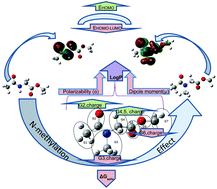
N-Methylation has a significant impact on improving the oral bioavailability, lipophilicity and aqueous solubility of peptide-based lead drug structures. The selected mono-amino acid derivatives Ac-X-OMe, where X = Gly, Val, Leu, Ile, Phe, Met, Cys, Ser, Asp and His as well as their corresponding N-methylated analogues were studied. The clog P values of all N-methylated peptides are greater than those of native compounds. Quantum chemical calculations were performed to estimate the aqueous solubility of these lipophilic compounds using density functional theory (DFT). To confirm the contribution of dispersion forces on quantum chemical data, the long-range corrected (LC) hybrid density functional (ωB97X-D) was also probed for some amino acid derivatives. The ωB97X functional gave similar results. Our results reveal that after mono N-methylation of the peptide backbone, ΔGsolv becomes more negative (more water soluble) while polarizability and dipole moment are also increased. Natural atomic charges derived by natural bond orbital (NBO) analysis of N, C, and O atoms involved in amide functional group become more positive/(less negative) after N-methylation. All N-methylated amino acids have higher EHOMO (less negative) in comparison with the amino acid analogues, and in all cases N-methylation decreases EHOMO–LUMO. The calculated amide cis/trans activation energies (EA) of all the N-methylated amino acid derivatives were lower than that of native species. N-methylation of these compounds leads to an increase in lipophilicity, aqueous solubility, polarization, dipole moment and lowering of the cis/trans amide energy barrier (EA).

 Please wait while we load your content...
Please wait while we load your content...