Alkynylation of steroids via Pd-free Sonogashira coupling†
Abstract
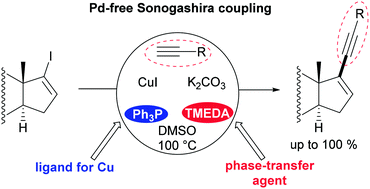
Cu-catalyzed Pd-free Sonogashira coupling has been proposed as a straightforward and convenient route to valuable steroidal enynes. A biligand catalyst system based on Ph3P and TMEDA has been designed. The protocol was utilized for the efficient coupling of iodosteroids with diverse terminal alkynes and 1-trimethylsilylalkynes. A possible role of an auxiliary ligand as a phase-transfer catalyst for a sparingly soluble inorganic base (K2CO3) was revealed.

 Please wait while we load your content...
Please wait while we load your content...