One-step protecting-group-free synthesis of azepinomycin in water†
Abstract
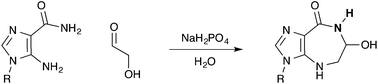
We report an efficient, atom economical general acid–base catalyzed one-step multi-gram synthesis of azepinomycin from commercially available compounds in water. We propose that the described pH-dependent Amadori rearrangement, which couples an amino-imidazole and simple sugar, is of importance as a potential step toward predisposed purine nucleotide synthesis at the origins of life.
- This article is part of the themed collection: Celebrating our 2019 Prize and Award winners

 Please wait while we load your content...
Please wait while we load your content...