Acid-promoted direct electrophilic trifluoromethylthiolation of phenols†
Abstract
The electrophilic aromatic ring trifluoromethylthiolation of various substituted phenols was accomplished using PhNHSCF3 (N-trifluoromethylsulfanyl)aniline, (1) in the presence of BF3·Et2O (2) or triflic acid as the promoter. The functionalization was exclusively para-selective; phenols unsubstituted in both the ortho- and para positions solely gave the para-substituted SCF3-products in all cases, while para-substituted phenols gave the ortho-substituted SCF3-products. 3,4-Dialkyl substituted phenols yielded the corresponding products according to the Mills-Nixon effect, and estrone and estradiol furnished biologically interesting SCF3-analogues. The highly reactive catechol and pyrogallol substrates gave the expected products smoothly in the presence of BF3·Et2O, whereas less reactive phenols required triflic acid. 2-Allylphenol gave the expected p-SCF3 analogue, which underwent an addition/cyclization sequence and furnished a new di-trifluoromethylthio substituted 2,3-dihydrobenzofuran derivative. Some additional transformations of 4-(trifluoromethylthio)phenol with NBS, NIS, HNO3, HNO3/H2SO4 and 4-bromobenzyl bromide were performed giving bromo-, iodo-, nitro- and benzyl substituted products. The latter derivative underwent Suzuki–Miyaura coupling with phenylboronic acid.
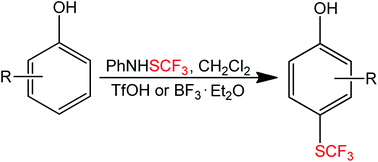

 Please wait while we load your content...
Please wait while we load your content...