Validating the alkene and alkyne hydrophosphonylation as an entry to organophosphonates
Abstract
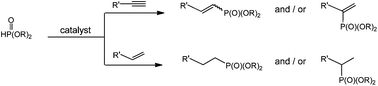
The first paper on the hydrophosphonylation of terminal alkenes was published in 1958 by Stiles and coworkers. Afterwards various papers described organometal-catalyzed and free-radical reactions leading to linear anti-Markovnikov adducts and/or branched Markovnikov products. In 1996 Han and Tanaka reported the first example of alkyne hydrophosphonylation catalyzed by a palladium complex. Further studies using other metal catalysts registered poor selectivity as mixtures of adducts were obtained in most of the cases examined. The first example of alkyne hydrophosphonylation by H-phosphonates under free-radical conditions leading to Z- and E-vinylphosphonates in a 1 : 1 ratio was reported by our group. Nevertheless, Z- to E-isomerization took place upon irradiation in the presence of a thiol.

 Please wait while we load your content...
Please wait while we load your content...