Rapid access to glycopeptide antibiotic precursor peptides coupled with cytochrome P450-mediated catalysis: towards a biomimetic synthesis of glycopeptide antibiotics†
Abstract
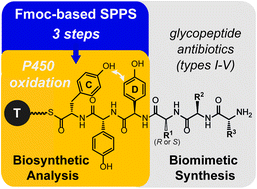
Understanding the mechanisms underpinning glycopeptide antibiotic biosynthesis is key to the future ability to reinvent these compounds. For effective in vitro characterization of the crucial later steps of the biosynthesis, facile access to a wide range of substrate peptides as their Coenzyme A (CoA) conjugates is essential. Here we report the development of a rapid route to glycopeptide precursor CoA conjugates that affords both high yields and excellent purities. This synthesis route is applicable to the synthesis of peptide CoA-conjugates containing racemization-prone arylglycine residues: such residues are hallmarks of non-ribosomal peptide synthesis and have previously been inaccessible to peptide synthesis using Fmoc-type chemistry. We have applied this route to generate glycopeptide precursor peptides in their carrier protein-bound form as substrates to explore the specificity of the first oxygenase enzyme from vancomycin biosynthesis (OxyBvan). Our results indicate that OxyBvan is a highly promiscuous catalyst for phenolic coupling of diverse glycopeptide precursors that accepts multiple carrier protein substrates, even on carrier protein domains from alternate glycopeptide biosynthetic machineries. These results represent the first important steps in the development of an in vitro biomimetic synthesis of modified glycopeptide aglycones.
 Please wait while we load your content...
Please wait while we load your content...