Synthesis, and QSAR analysis of anti-oncological active spiro-alkaloids†
Abstract
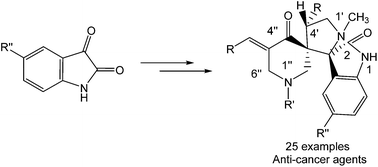
QSAR study describes the anti-neoplastic spiro-alkaloids with relevant molecular descriptors using CODESSA III software. The dispiro[3H-indole-3,2′-pyrrolidine-3′,3′′-piperidines] 24–48 were synthesized via [3 + 2]-cycloaddition reaction of azomethine ylides, (generated in situ via decarboxylative condensation of isatins 21–23 with sarcosine) and 3E,5E-1-alkyl-3,5-bis(arylmethylidene)-4-piperidones 10–20. Some of the synthesized analogues exhibited promising antitumor properties against HELA (cervical), HEPG2 (liver), T-47D, MCF7 (breast), and HCT116 (colon) human tumor cell lines, demonstrating activity close to or even better than the standard Doxorubicin, based on in vitro Sulfo-Rhodamine-B bio-assay.

 Please wait while we load your content...
Please wait while we load your content...