Synthesis of 1D-glyconanomaterials by a hybrid noncovalent–covalent functionalization of single wall carbon nanotubes: a study of their selective interactions with lectins and with live cells†
Abstract
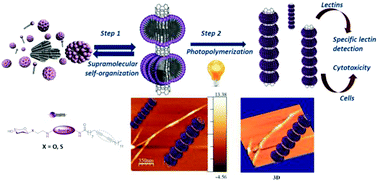
To take full advantage of the remarkable applications of carbon nanotubes in different fields, there is a need to develop effective methods to improve their water dispersion and biocompatibility while maintaining their physical properties. In this sense, current approaches suffer from serious drawbacks such as loss of electronic structure together with low surface coverage in the case of covalent functionalizations, or instability of the dynamic hybrids obtained by non-covalent functionalizations. In the present work, we examined the molecular basis of an original strategy that combines the advantages of both functionalizations without their main drawbacks. The hierarchical self-assembly of diacetylenic-based neoglycolipids into highly organized and compacted rings around the nanotubes, followed by photopolymerization leads to the formation of nanotubes covered with glyconanorings with a shish kebab-type topology exposing the carbohydrate ligands to the water phase in a multivalent fashion. The glyconanotubes obtained are fully functional, and able to establish specific interactions with their cognate receptors. In fact, by taking advantage of this selective binding, an easy method to sense lectins as a working model of toxin detection was developed based on a simple analysis of TEM images. Remarkably, different experimental settings to assess cell membrane integrity, cell growth kinetics and cell cycle demonstrated the cellular biocompatibility of the sugar-coated carbon nanotubes compared to pristine single-walled carbon nanotubes.

 Please wait while we load your content...
Please wait while we load your content...