Tridentate benzylthiols on Au(111): control of self-assembly geometry†
Abstract
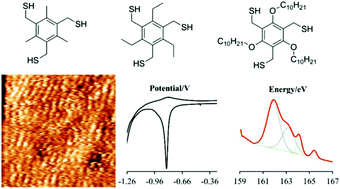
A set of hexasubstituted benzene derivatives with three thiol groups in the 1, 3, 5 positions and varied aliphatic substituents in the 2, 4, 6 positions (Me3-BTMT, Et3-BTMT, ODe3-BTMT) has been synthesized and self-assembled on Au(111). The resulting self-assembled monolayers (SAMs) are characterized by scanning tunneling microscopy (STM), X-ray photoelectron spectroscopy (XPS), and electrochemistry. The molecular orientation and long-range order are affected by the “gear effect” of the hexasubstituted benzene ring and van der Waals interactions between the physisorbed alkyl chains drive. Me3-BTMT adopts a standing up orientation which results in the highest molecular surface density but also the lowest degree of chemisorption (1 to 2 Au–S bonds per molecule). In contrast, Et3-BTMT favors a lying down orientation with a greater number of surface-bonded thiol groups (2 to 3) per molecule, associated with the peculiar geometry of this molecule. Finally, ODe3-BTMT adsorbs mainly in a lying down orientation, forming the SAM with the highest degree of chemisorption (all thiol groups are gold-bonded) and the lowest molecular areal density.

 Please wait while we load your content...
Please wait while we load your content...