Complexity in structure-directed prebiotic chemistry. Effect of a defective competing reactant in tetrapyrrole formation
Abstract
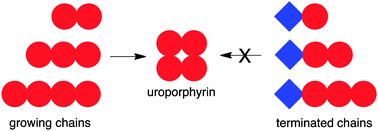
Chemical reactions in prebiotic environments likely entailed complex combinatorial processes with mixtures of reactants. As a case study, the effect of a defective reactant has been examined in a chemical model for the prebiogenesis of tetrapyrrole macrocycles (uroporphyrins). The latter are formed by a multistep process that includes (1) condensation of a β-diketone and an α-aminoketone to form a pyrrole, (2) self-condensation of the pyrrole to form a bilane (tetrapyrromethane), (3) cyclization of the bilane to form a porphyrinogen, and (4) oxidation of the porphyrinogen to form the porphyrin. A defective β-diketone (acetylacetone) affords a pyrrole that can react with nascent oligopyrromethane chains and in so doing terminate chain growth. Quantitative studies show that the porphyrin yield decreases with the fourth power of the mole fraction of the non-defective reactant. Mass spectral examination of the reaction mixture revealed the presence of the pyrrole and of end-capped oligomers derived from the defective diketone. Taken together, the results show the poisoning effect of a small amount of defective reactant in repetitive, irreversible structure-directed reactions, which have been a mainstay in thinking about chemistry leading to the origin of life.

 Please wait while we load your content...
Please wait while we load your content...