Enhanced simultaneous removal of malachite green and safranin O by ZnO nanorod-loaded activated carbon: modeling, optimization and adsorption isotherms
Abstract
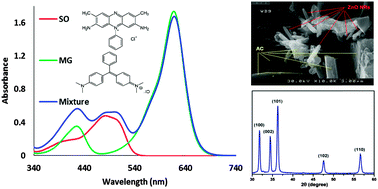
The simultaneous ultrasonic-assisted adsorption of malachite green (MG) and safranin O (SO) onto ZnO nanorod-loaded activated carbon (ZnO-NR-AC) as a green and safe nanostructured adsorbent was studied. The adsorbent was characterized by SEM, FT-IR and XRD. The dependence of adsorption efficiency on various parameters such as pH, sonication time, adsorbent mass, initial MG concentration and initial SO concentration was investigated by central composite design (CCD) under response surface methodology (RSM). A predictive model was successfully constructed for the suitable description of the real behavior of the adsorption state. The optimum conditions at which the maximum removal of MG (99%) and SO (95%) is achieved in a short time were obtained. The adsorption kinetics and equilibrium isotherm were shown to be well described by the pseudo-second-order kinetic and Langmuir models, respectively. Reasonably high values of 59.17 and 55.25 mg g−1 for the adsorption capacity of Zn-NR-AC were obtained for the adsorption of MG and SO, respectively.

 Please wait while we load your content...
Please wait while we load your content...