How the stereochemistry decides the selectivity: an approach towards metal ion detection†
Abstract
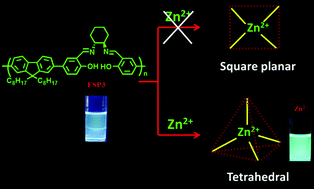
The stereochemistry of the coordination sites of a ligand plays a specific role in its binding with metals in a specific geometry. Herein, we designed and successfully prepared three different fluorene-based (A–B)n-type salen polymers (achiral FSP1, FSP2, and chiral FSP3), wherein the A-part is fluorophore and the B-part is the receptor. In the receptor, the coordination sites have four atoms (ONNO) that can bind any metal ion, but the orientation of ONNO differs in the three polymers. This orientation of the coordination site (i.e., the stereochemistry) into the receptor part of the polymer makes them more selective for a particular metal ion. In this study, it is shown that the orientation of the coordination sites of the receptor in the main chain polymer significantly determines the selective detection behavior for metal ions. Among the three polymers, FSP1 and FSP2 are sensitive towards different metal ions but are not selective towards any particular metal ion. However, in contrast, FSP3 is highly sensitive and selective to Zn2+ ions over other metal ions with a turn-on visible bright blue fluorescent color. This turn-on detection of the polymer is possibly due to the suppression of photo-induced electron transfer (PET) upon binding with Zn2+ ions. Theoretical calculations were also performed to show the orientation of the coordination sites. In FSP3, the coordination sites orient in a distorted tetrahedral fashion, which is very much prone to bind Zn2+ in a nearly tetrahedral geometry and that makes it more selective for Zn2+ ions only. The coordination geometry was also supported by 2D NMR studies. This report provides a template for the suitable design of a Zn2+ sensor, depending on the nature of the receptor incorporated into the main chain polymer.

 Please wait while we load your content...
Please wait while we load your content...