Lanthanum ion (La3+) substituted CoFe2O4 anode material for lithium ion battery applications†
Abstract
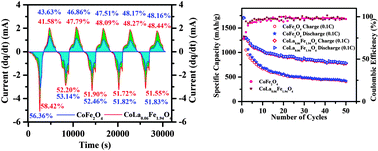
Mesoporous CoFe2O4 and lanthanum ion (La3+) substituted CoFe2O4 nanoparticles of composition CoLa0.06Fe1.94O4 were prepared by the citric acid and urea assisted combustion process and the citric acid, urea, and glycerol assisted foam combustion process, respectively. The effect of lanthanum ion substitution on the crystallite size, the lattice parameter, the particle morphology and electrical properties of pristine CoFe2O4 is studied using powder X-ray diffraction, Fourier transform infrared spectroscopy, scanning electron microscopy, transmission electron microscopy and impedance spectroscopy. For electrochemical studies, half cells are fabricated in CR 2032 coin cells using mesoporous CoFe2O4 and CoLa0.06Fe1.94O4 as anode materials. The charge/discharge performances of half cells are studied using cyclic voltammetry and charge/discharge cycling. The rare earth ion substitution improved the structural stability of pristine CoFe2O4 and enhanced the oxidation reaction which maintained the number of lithium ions involved in the oxidation and reduction reactions. It also improved the battery performance, for instance, the specific capacity of La3+-ion doped CoFe2O4 is 188% compared to the pristine CoFe2O4 anode even after 50 cycles.

 Please wait while we load your content...
Please wait while we load your content...