Thermodynamically feasible photoelectron transfer from bioactive π-expanded imidazole luminophores to ZnO nanocrystals†
Abstract
The photoinduced electron transfer (PET) based sensor, 2-(1-(naphthalene-1-yl)-4,5-diphenyl-1H-imidazol-2-yl)phenol, was synthesised and characterized by spectral techniques. Absorption, emission, lifetime and cyclic voltammetric studies have been made to probe the binding interaction between imidazole and ZnO nanocrystals. The nanocrystalline ZnO enhances the absorbance and emission intensity of imidazole and the binding constant for the formation of the imidazole–ZnO composite has been obtained. The band gap energy of ZnO is greater than the excited state energy of imidazole and the emission enhancement is only due to the electron transfer process. TEM, SEM and EDX spectra confirmed the binding of imidazole with nanocrystalline ZnO. Theoretical investigation reveals that small ZnnOn clusters (n < 9) and their imidazole–ZnO composites are stabilized in 2D ring geometries whereas the larger cluster Zn10O10 and its imidazole–ZnO composite prefers 3D cage structures. The ring to cage crossover of ZnO clusters is studied by analyzing the Zn–Ô–Zn and 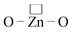 bond angles, Zn–O bond length and number of bonds. Binding energy, energy gap, binding site and adsorption strength of imidazole with different ZnO clusters show that Zn2+ of ZnO clusters prefers to bind with the azomethine nitrogen atom (N-site) relative to other binding sites (O-site). The synthesized imidazole is an efficient fluorescent chemosensor for Zn2+ ions; it binds with Zn2+ ions in aqueous alcohol strongly to exhibit the sensing behaviour.
bond angles, Zn–O bond length and number of bonds. Binding energy, energy gap, binding site and adsorption strength of imidazole with different ZnO clusters show that Zn2+ of ZnO clusters prefers to bind with the azomethine nitrogen atom (N-site) relative to other binding sites (O-site). The synthesized imidazole is an efficient fluorescent chemosensor for Zn2+ ions; it binds with Zn2+ ions in aqueous alcohol strongly to exhibit the sensing behaviour.


 Please wait while we load your content...
Please wait while we load your content...