From Ni-based nanoprecursors to NiO nanostructures: morphology-controlled synthesis and structure-dependent electrochemical behavior
Abstract
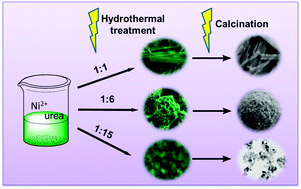
Phase transformation and morphology tailoring of NiO nanostructures have been successfully achieved using Ni-based precursors (Ni(SO4)0.3(OH)1.4 and Ni(HCO3)2) synthesized by a facile urea-controlled hydrothermal route. The as-synthesized NiO nanomaterials were characterized by X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), and nitrogen adsorption analyses. The electrochemical performance of the as-prepared NiO nanostructures was evaluated by CV and galvanostatic charge–discharge tests. NiO nanoparticles with larger surface areas exhibited higher electrochemical performance than nanobelts and nanospheres when used as the supercapacitor electrodes. The discharge capacity of NiO nanoparticles can retain 609.5 F g−1 after 500 cycles at a charging–discharging current density of 5 A g−1.

 Please wait while we load your content...
Please wait while we load your content...