Novel one-pot green synthesis of graphene in aqueous medium under microwave irradiation using a regenerative catalyst and the study of its electrochemical properties†
Abstract
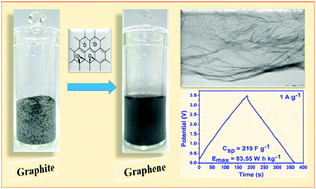
In this work we report an economic, eco-friendly, high yielding and facile one-pot method for the large scale synthesis of few layer graphene (FLG) nanosheets directly from graphite in aqueous medium using a regenerative catalyst, sodium tungstate. This method is fast and makes use of environmental friendly chemicals and microwave radiation. The as-synthesized FLG nanosheets are characterized by field emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), X-ray diffraction (XRD), Raman spectroscopy, X-ray photoelectron spectroscopy (XPS) and Brunauer–Emmett–Teller (BET) surface area analysis. Raman analysis indicates that the as-synthesized graphene is bilayered with a smaller domain size of 3.9 nm which is responsible for a higher specific surface area of FLG nanosheets (1103.62 m2 g−1). Moreover, XPS analysis of FLG nanosheets shows a high C : O ratio (∼9.6) which is the best among the graphene prepared from green chemicals. The electrochemical performance of as-synthesized FLG nanosheets is analysed by cyclic voltammetry (CV), chronopotentiometry and electrochemical impedance spectroscopy (EIS) in neat 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIMBF4) electrolyte. The superior capacitive performance with large capacitance (219 F g−1), high energy density (83.56 W h kg−1) and excellent cyclability (3000 cycles) exhibited by these graphene nanosheets make them an excellent candidate for supercapacitor material.

 Please wait while we load your content...
Please wait while we load your content...