On-line coupling of continuous-flow gel electrophoresis with inductively coupled plasma-mass spectrometry to quantitatively evaluate intracellular metal binding properties of metallochaperones HpHypA and HpHspA in E. coli cells†
Abstract
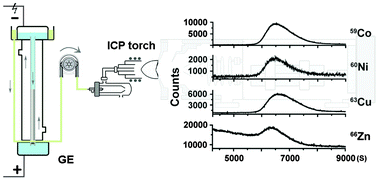
On-line coupling of gel electrophoresis with inductively coupled plasma-mass spectrometry (GE-ICP-MS) offers a strategy to monitor intracellular metals and their associated proteins simultaneously. Herein, we examine the feasibility of the GE-ICP-MS system in the quantitative analysis of intracellular metal binding properties using two Helicobacter pylori metallochaperones HypA and HspA overexpressed in E. coli cells as showcases. We show that parallel detection of metal and sulfur signals allows accurate quantification of intracellular metal–protein stoichiometries, even for metalloproteins that bind metal ions with micromolar affinities. Using this approach, we demonstrate that only a trace amount of Ni2+ is associated with HpHypA in cells, distinct from the in vitro observation of stoichiometric binding, while HpHypA exhibits high fidelity towards its structural metal Zn2+ with stoichiometric Zn2+ binding. In contrast, HpHspA associates with Zn2+, Ni2+, Cu2+ and Co2+ from an essential metal pool with ca. 0.5 molar equivalents of total metals bound per HpHspA monomer. The metal binding properties of both HpHypA and HpHspA were altered by Bi3+. The binding of both Zn2+ and Ni2+ to HpHypA was suppressed under the stress of Bi3+ in cells, different from in vitro studies that showed that Bi3+ interfered with Zn2+ but not Ni2+ binding. This study provides an analytical approach to investigate the intracellular metal selectivity of overexpressed metalloproteins.

 Please wait while we load your content...
Please wait while we load your content...