Antimicrobial spine-bone cement with caffeic acid phenethyl ester for controlled release formulation and in vivo biological assessments†
Abstract
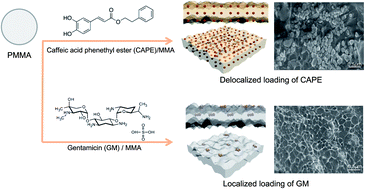
The work reports the preparation of antimicrobial spine-bone-cement with a highly controlled release formulation that was subjected to tests, involving in vitro and in vivo biological assessments such as antimicrobial effects, a cytotoxicity test, a bacterial reverse mutation (Ames) assay, a micronucleus assay, and implantation analysis. Two kinds of acrylic bone-cements were tested for the expression of antimicrobial and antibiotic activity. For these tests, new antimicrobial bone-cement was prepared using a caffeic acid phenethyl ester (CAPE) compound from natural propolis, and an antibiotic bone-cement was prepared by the inclusion of gentamycin (GM). The antimicrobial ability and controlled release property of both CAPE-loaded and GM-loaded bone-cements were demonstrated as a function of time, concentration and optimization to maximize their antimicrobial effects against Staphylococcus aureus bacteria. The compressive strengths of both bone-cements, at various CAPE and GM concentrations, were over 130 MPa and 80 MPa for CAPE-and GM-loaded bone-cement, respectively. Moreover, the CAPE-loaded antimicrobial bone-cements showed a controlled release pattern better than that of the GM-loaded antibiotic bone-cement due to their homogeneous loading. Through various in vitro and in vivo biological assessments, the CAPE-loaded antimicrobial bone-cements showed positive results in all assessments, without any inflammatory response.

 Please wait while we load your content...
Please wait while we load your content...