Production of long chain alcohols and alkanes upon coexpression of an acyl-ACP reductase and aldehyde-deformylating oxygenase with a bacterial type-I fatty acid synthase in E. coli†
Abstract
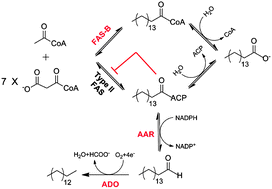
Microbial long chain alcohols and alkanes are renewable biofuels that could one day replace petroleum-derived fuels. Here we report a novel pathway for high efficiency production of these products in Escherichia coli strain BL21(DE3). We first identified the acyl-ACP reductase/aldehyde deformylase combinations with the highest activity in this strain. Next, we used catalase coexpression to remove toxic byproducts and increase the overall titer. Finally, by introducing the type-I fatty acid synthase from Corynebacterium ammoniagenes, we were able to bypass host regulatory mechanisms of fatty acid synthesis that have thus far hampered efforts to optimize the yield of acyl-ACP-derived products in BL21(DE3). When all these engineering strategies were combined with subsequent optimization of fermentation conditions, we were able to achieve a final titer around 100 mg L−1 long chain alcohol/alkane products including a 57 mg L−1 titer of pentadecane, the highest titer reported in E. coli BL21(DE3) to date. The expression of prokaryotic type-I fatty acid synthases offer a unique strategy to produce fatty acid-derived products in E. coli that does not rely exclusively on the endogenous type-II fatty acid synthase system.

 Please wait while we load your content...
Please wait while we load your content...