Low complexity and disordered regions of proteins have different structural and amino acid preferences†
Abstract
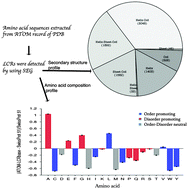
Low complexity regions (LCRs) or non-random regions of a few amino acids are abundantly present in proteins. LCRs are traditionally considered as floppy structures with high solvent accessibility. Thus little attention was paid to them for structural studies. However LCRs have been found to contain information relevant to protein structure and various important functions. The present study is an attempt to understand the structural trend of LCRs. Here we report a study conducted to understand the structural trend, solvent accessibility and amino acid preferences of LCRs. The results show that LCRs might attain any type of secondary structure; however, the helix is frequently seen, whereas sheets occur rarely. We also found that LCRs are not always exposed on the surface. We found insignificant contribution of trans-membrane helices to the overall helix content. The LCRs having a secondary structure have different enrichment and depletion of amino acids from LCRs without a secondary structure and disordered protein sequences. However, LCRs of NMR structures showed compositional and functional similarity to the disordered regions of proteins. We also noted that in ∼3/4 LCRs, the entire amino acid did not have a single structural class, but rather an ensemble of more than one secondary structure, which indicates that they are found at places where structure transition occurs. Overall analysis suggests that the overall protein sequence has a greater influence on the structural and sequence enrichment rather than only the local amino acid composition of LCRs.

 Please wait while we load your content...
Please wait while we load your content...