Evaluation of photodynamic therapy efficiency using an in vitro three-dimensional microfluidic breast cancer tissue model
Abstract
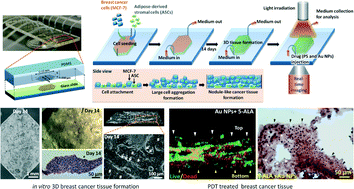
In recognition of the limitations of monolayer cell cultures and resource-intensive animal studies, a microfluidic culture system was developed for creation of physiologically relevant three-dimensional (3D) tissues. In this study, an in vitro 3D breast cancer tissue model was established in a microfluidic system with human breast cancer cells (MCF-7) and primary adipose-derived stromal cells (ASCs). It was evaluated for utility in determining the efficiency of photodynamic therapy (PDT) with therapeutic agents (i.e. photosensitizer and gold nanoparticles) under various irradiation conditions. We demonstrated, for the first time, the potential use of a microfluidic-based in vitro 3D breast cancer model for effective evaluation of PDT, with the capability of controlling 3D microenvironments for breast cancer tissue formation, real-time monitoring of tissue progression, implementing a circulation-like dynamic medium flow and drug supplements, and investigating the relation between light penetration and tissue depth in PDT.

 Please wait while we load your content...
Please wait while we load your content...