A droplet-to-digital (D2D) microfluidic device for single cell assays†
Abstract
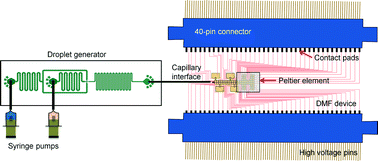
We have developed a new hybrid droplet-to-digital microfluidic platform (D2D) that integrates droplet-in-channel microfluidics with digital microfluidics (DMF) for performing multi-step assays. This D2D platform combines the strengths of the two formats—droplets-in-channel for facile generation of droplets containing single cells, and DMF for on-demand manipulation of droplets including control of different droplet volumes (pL–μL), creation of a dilution series of ionic liquid (IL), and parallel single cell culturing and analysis for IL toxicity screening. This D2D device also allows for automated analysis that includes a feedback-controlled system for merging and splitting of droplets to add reagents, an integrated Peltier element for parallel cell culture at optimum temperature, and an impedance sensing mechanism to control the flow rate for droplet generation and preventing droplet evaporation. Droplet-in-channel is well-suited for encapsulation of single cells as it allows the careful manipulation of flow rates of aqueous phase containing cells and oil to optimize encapsulation. Once single cell containing droplets are generated, they are transferred to a DMF chip via a capillary where they are merged with droplets containing IL and cultured at 30 °C. The DMF chip, in addition to permitting cell culture and reagent (ionic liquid/salt) addition, also allows recovery of individual droplets for off-chip analysis such as further culturing and measurement of ethanol production. The D2D chip was used to evaluate the effect of IL/salt type (four types: NaOAc, NaCl, [C2mim] [OAc], [C2mim] [Cl]) and concentration (four concentrations: 0, 37.5, 75, 150 mM) on the growth kinetics and ethanol production of yeast and as expected, increasing IL concentration led to lower biomass and ethanol production. Specifically, [C2mim] [OAc] had inhibitory effects on yeast growth at concentrations 75 and 150 mM and significantly reduced their ethanol production compared to cells grown in other ILs/salts. The growth curve trends obtained by D2D matched conventional yeast culturing in microtiter wells, validating the D2D platform. We believe that our approach represents a generic platform for multi-step biochemical assays such as drug screening, digital PCR, enzyme assays, immunoassays and cell-based assays.

 Please wait while we load your content...
Please wait while we load your content...