Evaluation of sample preparation methods for single cell quantitative elemental imaging using proton or synchrotron radiation focused beams
Abstract
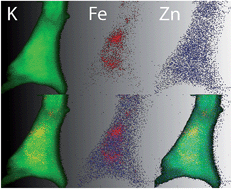
Particle induced X-ray emission with a focused beam (μPIXE) and synchrotron-based X-ray micro-fluorescence (μSXRF) are used to determine the distribution and contents of trace elements in single cells. A proper sample preparation method is required to ensure that the elemental distribution is preserved spatially and quantitatively. The aim of this study was to establish an optimal sample preparation method for single whole cell microanalysis, compatible with both μPIXE and μSXRF techniques. To find the most efficient method, we used PC12 cells as the cellular model and compared four widely applied protocols using a combination of rinsing solutions (phosphate buffered saline or ammonium acetate) and fixation methods (cryofixation or chemical fixation with 3% paraformaldehyde or methanol). The results showed a loss of diffusible elements K and Mg and an increase in Na, S, Cl and Zn concentrations in chemically fixed cells compared to cryofixed cells. In addition, K/Na and Cl/K cellular ratios indicated a good preservation of the chemical and structural integrity of cryofixed cells but not those of chemically fixed ones. The disturbance of elemental distributions after chemical fixation was also observed on rat brain tissue sections. Our results suggest that the optimal sample preparation method to study elemental distribution in single whole cells prepared for X-ray microanalysis is achieved when cells are rinsed with ammonium acetate, quickly frozen by plunging into liquid nitrogen–chilled cryogenic fluid and freeze-dried at low temperatures. This protocol was also successfully validated on rat primary hippocampal neurons, a delicate in vitro neuronal model.

 Please wait while we load your content...
Please wait while we load your content...