Development of an isolation procedure and MC-ICP-MS measurement protocol for the study of stable isotope ratio variations of nickel†
Abstract
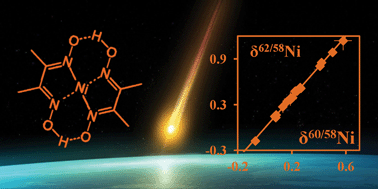
Variations in the isotopic composition of Ni resulting from natural mass-dependent processes in terrestrial or extraterrestrial conditions, inhomogeneous distribution of nucleosynthetic components and/or ingrowth from radioactive parent nuclides, help us to further understand the early formation history of Solar System materials and the nature of the processes these materials subsequently experienced. In studies of Ni isotope systematics, mass-dependent variations in the isotopic composition of Ni are often bypassed because of the challenges associated with the sample preparation. At the level of natural variation studied, Ni isotope ratio measurements are extremely sensitive to spectral interference, artificial on-column isotope fractionation and possibly even to the mass bias correction model applied. To adequately address these complications, an isolation procedure and measurement protocol relying on multi-collector ICP-mass spectrometry (MC-ICP-MS) have been designed and validated in this work. The overall reproducibility obtained based on repeated measurement of a Sigma-Aldrich high-purity Ni standard is 0.036‰, 0.049‰, 0.078‰ and 0.53‰ for δ60/58Ni, δ61/58Ni, δ62/58Ni and δ64/58Ni, respectively (n = 14; 2 SD). Nickel isotope ratio variations have been studied in a set of iron meteorites and geological reference materials, and the results obtained, except for those suffering from an elevated 64Zn background, show good agreement with the available literature data. By using the flexible generalized power law with a variable discrimination exponent and the three-isotope method, the processes underlying natural mass fractionation of Ni for terrestrial reference materials were found to have a mixed equilibrium/kinetic nature. Mass-dependent Ni fractionation was observed between sample fractions of the Canyon Diablo iron meteorite, and the extracted fractionation factor β corresponds to isotope partitioning following the power law.
- This article is part of the themed collection: JAAS Emerging Investigator Lectureship winners

 Please wait while we load your content...
Please wait while we load your content...