A microfluidic platform reveals differential response of regulatory T cells to micropatterned costimulation arrays†
Abstract
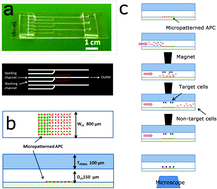
T cells are key mediators of adaptive immunity. However, the overall immune response is often directed by minor subpopulations of this heterogeneous family of cells, owing to specificity of activation and amplification of functional response. Knowledge of differences in signaling and function between T cell subtypes is far from complete, but is clearly needed for understanding and ultimately leveraging this branch of the adaptive immune response. This report investigates differences in cell response to micropatterned surfaces by conventional and regulatory T cells. Specifically, the ability of cells to respond to the microscale geometry of TCR/CD3 and CD28 engagement is made possible using a magnetic-microfluidic device that overcomes limitations in imaging efficiency associated with conventional microscopy equipment. This device can be readily assembled onto micropatterned surfaces while maintaining the activity of proteins and other biomolecules necessary for such studies. In operation, a target population of cells is tagged using paramagnetic beads, and then trapped in a divergent magnetic field within the chamber. Following washing, the target cells are released to interact with a designated surface. Characterization of this system with mouse CD4+ T cells demonstrated a 50-fold increase in target-to-background cell purity, with an 80% collection efficiency. Applying this approach to CD4+CD25+ regulatory T cells, it is then demonstrated that these rare cells respond less selectively to micro-scale features of anti-CD3 antibodies than CD4+CD25− conventional T cells, revealing a difference in balance between TCR/CD3 and LFA-1-based adhesion. PKC-θ localized to the distal pole of regulatory T cells, away from the cell–substrate interface, suggests a mechanism for differential regulation of TCR/LFA-1-based adhesion. Moreover, specificity of cell adhesion to anti-CD3 features was dependent on the relative position of anti-CD28 signaling within the cell–substrate interface, revealing an important role for coincidence of TCR and costimulatory pathway in triggering regulatory T cell function.

 Please wait while we load your content...
Please wait while we load your content...