Actin cytoskeleton differentially alters the dynamics of lamin A, HP1α and H2B core histone proteins to remodel chromatin condensation state in living cells†
Abstract
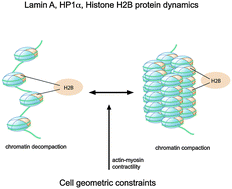
Cells in physical microenvironments regulate their functioning and geometry in response to mechanical stimuli. Recent studies have demonstrated the influence of the integrated actin cytoskeleton on nuclear integrity and chromatin organization. However, the mechanisms underlying the mechanotransduction of their physical coupling to nuclear protein dynamics are not well understood. In this study, we take advantage of micropatterned geometric substrates in NIH3T3 mouse fibroblasts to probe the functional influence of actin organization on nuclear lamina and chromatin assembly. Fluorescence correlation spectroscopy studies demonstrate that stabilization of perinuclear actin strengthens the transient interactions of lamin A with the chromatin. Correspondingly, fluorescence recovery after photobleaching studies reveal enhanced mobility of these nuclear lamina proteins when actin organization is perturbed. Combining these fluorescence dynamics assays, we also demonstrate an actin-driven differential modulation of core histone H2B and heterochromatin HP1α protein dynamics with chromatin. These altered dynamics are reflected structurally by concomitant changes in the architecture of the heterochromatin foci as seen by immunofluorescence assays. Taken together, our study provides a demonstration of the differential mechanical control of perinuclear actin on the dynamics of the nuclear lamina, euchromatin and heterochromatin regimes of the nucleus, and suggests an actin-mediated route to spatially and structurally tune chromatin organization and dynamics.
- This article is part of the themed collection: Mechanobiology

 Please wait while we load your content...
Please wait while we load your content...