CO2 methanation over heterogeneous catalysts: recent progress and future prospects
Abstract
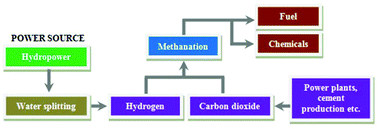
Catalytic approaches for CO2 fixation can play an important role, because CO2 can be artificially converted into reusable chemicals. Among the catalytic reactions of CO2, hydrogenation of CO2, the so-called methanation reaction, is a suitable technique for the fixation of CO2. This technique can be used to convert exhausted CO2 into methane (CH4), which can be recycled for use as a fuel or a chemical as well as by contributing to the reduction of CO2 emissions. Heterogeneous catalysts have been used to hydrogenate CO2 to methane. Heterogeneous catalysts are preferable to homogenous catalysts in terms of stability, separation, handling, and reuse, which reflects in lower costs for large-scale production. Significant progress has been made in this direction, the exploitation of novel heterogeneous catalysts. In this review, we discussed recent developments in this area, with emphases on catalytic reactivity and its physicochemical properties and reaction mechanism. Apart from materials aspects and catalytic performance, we also discuss fundamental strategies for the rational design of materials for effective transformation of CO2 to methane with the help of H2 and power sources.
- This article is part of the themed collection: 2015 most accessed Green Chemistry articles

 Please wait while we load your content...
Please wait while we load your content...