Structure-dependent tautomerization induced catalyst-free autocatalyzed N-alkylation of heteroaryl amines with alcohols†
Abstract
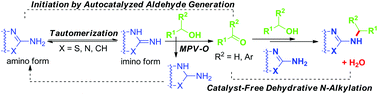
Catalyst-free autocatalyzed dehydrative N-alkylation reactions of 2-aminobenzothiazoles, 2-aminopyrimidines, and 2-aminopyrazine with primary and secondary alcohols have been achieved for efficient, practical, and green synthesis of the versatile heteroaryl amine derivatives. These reactions were interestingly induced by structure-dependent tautomeric equilibria of the heteroaryl amines via MPV–O transfer hydrogenation of the imino tautomers by alcohols to give aldehydes as the key initiating step.

 Please wait while we load your content...
Please wait while we load your content...