Continuous catalytic upgrading of ethanol to n-butanol and >C4 products over Cu/CeO2 catalysts in supercritical CO2†
Abstract
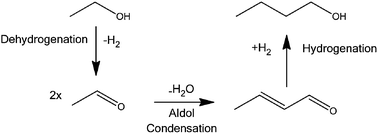
n-Butanol (BuOH) often has superior properties as a bio-fuel compared to ethanol (EtOH). However finding sustainable sources of BuOH is proving difficult. In this paper, direct production of BuOH from EtOH is compared over custom-synthesized six Cu catalysts, supported on different solid acids. These catalysts were tested in a continuous flow supercritical CO2 (scCO2) reactor, and were found to catalyse the dehydrogenation, aldol condensation and hydrogenation steps of the so-called Guerbet reaction converting EtOH to BuOH. BuOH yields and selectivities were significantly different over the four catalysts. Cu on high surface area CeO2 showed the best activity for BuOH formation, with yields above 30% achieved with good selectivity. In addition high pressure CO2 is shown to have a positive effect on the reaction, possibly due to the redox cycle of Ce2O3 and CeO2.
- This article is part of the themed collection: Celebrating the 2016 RSC Prize and Award Winners

 Please wait while we load your content...
Please wait while we load your content...