Electrochemical silver dissolution and recovery as a potential method to disinfect drinking water for underprivileged societies
Abstract
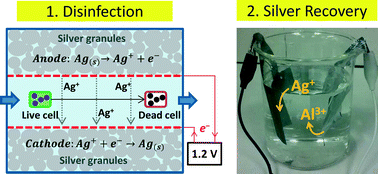
Limited access to disinfected drinking water is a critical challenge in developing countries. Ionic silver has biocidal capabilities and silver ionization is an established method for cooling water disinfection. However, silver ionization cannot be used in drinking water treatment because of its inherent human health risks. In this proof-of-concept study, a laboratory scale electrochemical reactor was developed where ionic silver is released via oxidation of metallic silver and subsequently recovered through cathodic reduction. Complete inactivation of Escherichia coli was demonstrated in 30 min at an application of 1.2 V. A high silver concentration (>1 mg L−1) was achieved in less than two minutes of water residence time within the disinfection reactor. The effluent silver concentration was linearly proportional to the electric current, allowing reliable estimation of silver concentration and corresponding disinfection performance by measuring electric current using an electrical multimeter. The disinfected water was further treated in a separate silver recovery cell. The silver concentration dropped down to safe levels for continued human consumption (0.1 mg L−1) via spontaneous cathodic silver reduction coupled with anodic oxidation of metallic aluminum. The estimated energy requirement was 6.1 kJ to treat 1 m3 of water; thus, solar panels or bicycle generators can provide sufficient energy for disinfection. The electrochemical disinfection and subsequent silver recovery methods have a strong potential as a prospective method to supply safe drinking water in underprivileged and remote societies.

 Please wait while we load your content...
Please wait while we load your content...