Biocatalytic perchlorate reduction: kinetics and effects of groundwater characteristics
Abstract
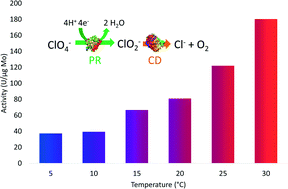
Biocatalytic reduction of perchlorate can minimize the effects of competitive electron acceptors and completely reduce perchlorate into chloride and oxygen, but to date has only been demonstrated under idealized laboratory conditions. This work investigated biocatalytic perchlorate reduction in two groundwater drinking water sources, under a range of conditions and with a variety of electron donors. The biocatalysts, perchlorate reductase and chlorite dismutase from Azospira oryzae, had a maximum activity of 162.5 ± 8.4 U (μg Mo)−1 in buffered solution and retained 82–94% of their activity in groundwater samples. The half saturation concentration for perchlorate was 92.0 μM. Perchlorate reduction rates were higher than nitrate reduction rates, with nitrate as the sole electron acceptor having reduction rates 7.5 to 9.7% of the maximum perchlorate reduction rates in groundwater. Activity was consistent from pH 6.5 to 9.0. The temperature dependence of biocatalytic perchlorate reduction was well defined by the Arrhenius equation. No significant difference in biocatalytic activity was observed with calcium and magnesium concentrations over the tested range of 0 to 400 mg L−1 or with natural organic matter up to 6 mg L−1. Ascorbic acid with addition of an electron shuttle resulted in reduction of more than 99% of perchlorate in less than 6 hours, an order of magnitude loss in activity compared to methyl viologen. These results suggest the potential of the biocatalysts for treating perchlorate over a range of concentrations and conditions representative of industrial and groundwater perchlorate contamination.


 Please wait while we load your content...
Please wait while we load your content...