Electrochemical wastewater treatment with carbon nanotube filters coupled with in situ generated H2O2†
Abstract
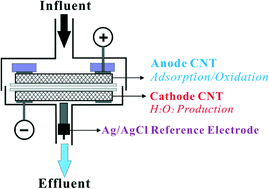
Electrochemically active carbon nanotube (CNT) filters can effectively adsorb and oxidize chemical compounds in the anode, but the role of a cathode in electrochemical filters beyond a counter electrode has not been thoroughly investigated. In this study, a novel wastewater treatment system was developed to combine both adsorption and oxidation in the CNT anode and additional oxidation with in situ generated hydrogen peroxide (H2O2) in the CNT cathode. The impacting factors, treatment efficiency, and oxidation mechanism of the system were systematically studied. The results demonstrated that H2O2 flux could be affected by the electrode material, cathode potential, pH, flow rate, and dissolved oxygen (DO). The maximum H2O2 flux of 1.38 mol L−1 m−2 was achieved with C-grade CNT at an applied cathode potential of −0.4 V (vs. Ag/AgCl), a pH of 6.46, a flow rate of 1.5 mL min−1, and an influent DO flux of 1.95 mol L−1 m−2. Additionally, phenol was used as a model aromatic compound to evaluate the removal efficiency of the system and its oxidation rate was directly correlated with H2O2 flux. H2O2 was likely reacting with a phenol species that was anodically activated to a radical form, since H2O2 alone cannot remove phenol efficiently. Furthermore, electrochemical polymer formation via phenolic radical chain reactions may also contribute to 13% of phenol removal. A stable phenol removal efficiency of 87.0 ± 1.8% within 4 h of continuous operation was achieved with an average oxidation rate of 0.059 ± 0.001 mol h−1 m−2. The developed electrochemical CNT filtration system coupled with in situ generated H2O2 is a new application of carbon nanotube filters and can be used as an effective wastewater treatment system to remove organic pollutants or as a promising point-of-use wastewater treatment system.

 Please wait while we load your content...
Please wait while we load your content...