A quantitative analysis of the efficiency of solar-driven water-splitting device designs based on tandem photoabsorbers patterned with islands of metallic electrocatalysts†
Abstract
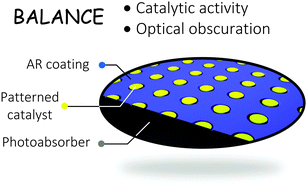
The trade-off between the optical obscuration and kinetic overpotentials of electrocatalyst films patterned onto the surface of tandem light-absorber structures in model photoelectrosynthetic water-splitting systems was investigated using a 0-dimensional load-line analysis and experimental measurements. The electrocatalytic performance of the catalyst at high current densities, normalized to the electrocatalyst surface area, is an important factor in the dependence of the optimal solar-to-hydrogen (STH) conversion efficiency, ηSTH,opt, on the filling fraction (fc) of the patterned catalysts, because even under conditions that produce minority-carrier current densities of ∼10 mA cm−2 at the solid/liquid interface, the current density at catalyst-bearing sites can be >1–2 A cm−2 in low filling-fraction films. A universal current-density versus potential relationship, up to current densities of 10 A cm−2, was obtained experimentally for the hydrogen-evolution reaction (HER) using patterned Pt ultramicroelectrode (UME) arrays with a range of filling fractions and disc diameters. The ηSTH,opt of system designs that utilize patterned electrocatalysts located on the illuminated side of tandem photoabsorbers was then evaluated systematically. The maximum STH conversion efficiency, ηSTH,max, using a hypothetical electrocatalyst that was optically transparent but which nevertheless exhibited a current-density versus potential behavior that is characteristic of the most active Pt films measured experimentally regardless of their optical obscuration, was 26.7%. By comparison, the maximum ηSTH,opt of 24.9% for real patterned Pt electrocatalyst films closely approached this ideal-case limit. The performance and materials utilization of the patterned electrocatalysts and of the uniformly coated electrocatalysts on tandem photoabsorbers were also compared in this study. Hence, patterned electrocatalysts with very low filling fractions can provide a potentially promising path to the realization of efficient large-scale photoelectrolysis systems while minimizing the use of scarce noble metals.

 Please wait while we load your content...
Please wait while we load your content...