N-doped TiO2 nanotubes coated with a thin TaOxNy layer for photoelectrochemical water splitting: dual bulk and surface modification of photoanodes†
Abstract
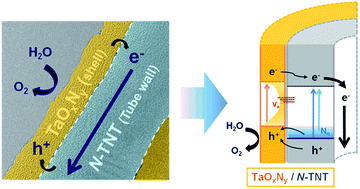
TaON is a good photoanode material with a suitable band structure for water splitting as well as coupling with TiO2 for efficient charge separation. However, the synthesis of TaON that requires high temperature nitridation (850 °C) limits the combination with other materials. In this work, we deposited a thin amorphous TaOxNy layer on N-doped TiO2 nanotubes (N-TNTs) through low temperature nitridation (500 °C) and demonstrated its successful performance as an efficient photoanode for water-splitting. Since the preparation temperature is low, TaOxNy on N-TNTs has a unique amorphous structure with a smooth thin layer (5 nm). It is proposed that the thin amorphous TaOxNy layer plays dual roles: (i) surface sensitization and/or charge rectification at the heterojunction between the TaOxNy layer and N-TNTs, and (ii) passivation of N-TNT surface trap states to retard the charge recombination. TaOxNy layer-decorated N-TNTs as dual modified TNTs (N-doping in the bulk and TaOxNy overlayer deposition on the surface) have significantly improved both visible (ca. 3.6 times) and UV (ca. 1.8 times) activities for PEC water-splitting as well as the faradaic efficiency (ca. 1.4 times, η = 98%) for H2 production. Making the amorphous TaOxNy layer crystalline at higher temperatures reduced the PEC activity of the hybrid photoanode, in contrast, which indicates that the amorphous TaOxNy layer deposition on N-TNTs through low temperature nitridation (500 °C) is optimized for the PEC activity. A range of spectroscopic and electrochemical techniques were systematically employed to investigate the properties of the PEC process.
- This article is part of the themed collection: 2020 EES Lectureship Winner: Wooyul Kim

 Please wait while we load your content...
Please wait while we load your content...