Abstract
Tin(II) ketoacidoximates of the type [HON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CRCOO]2Sn (R = Me 1, CH2Ph 2) and (MeON
CRCOO]2Sn (R = Me 1, CH2Ph 2) and (MeON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CMeCOO)3Sn]− NH4+·2H2O 3 were synthesized by reacting pyruvate- and hydroxyl- or methoxylamine RONH2 (R = H, Me) with tin(II) chloride dihydrate SnCl2·2H2O. The single crystal X-ray structure reveals that the geometry at the Sn atom is trigonal bipyramidal in 1, 2 and trigonal pyramidal in 3. Inter- or intramolecular hydrogen bonding is observed in 1–3. Thermogravimetric (TG) analysis shows that the decomposition of 1–3 to SnO occurs at ca. 160 °C. The evolved gas analysis during TG indicates complete loss of the oximato ligand in one step for 1 whereas a small organic residue is additionally removed at temperatures >400 °C for 2. Above 140 °C, [HON
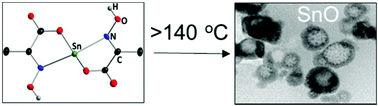
CMeCOO)3Sn]− NH4+·2H2O 3 were synthesized by reacting pyruvate- and hydroxyl- or methoxylamine RONH2 (R = H, Me) with tin(II) chloride dihydrate SnCl2·2H2O. The single crystal X-ray structure reveals that the geometry at the Sn atom is trigonal bipyramidal in 1, 2 and trigonal pyramidal in 3. Inter- or intramolecular hydrogen bonding is observed in 1–3. Thermogravimetric (TG) analysis shows that the decomposition of 1–3 to SnO occurs at ca. 160 °C. The evolved gas analysis during TG indicates complete loss of the oximato ligand in one step for 1 whereas a small organic residue is additionally removed at temperatures >400 °C for 2. Above 140 °C, [HON![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Me)COO]2Sn (1) decomposes in air to spherical SnO particles of size 10–500 nm. Spin coating of 1 on Si or a glass substrate followed by heating at 200 °C results in a uniform film of SnO. The band gap of the produced SnO film and nanomaterial was determined by diffuse reflectance spectroscopy to be in the range of 3.0–3.3 eV. X-ray photoelectron spectroscopy indicates surface oxidation of the SnO film to SnO2 in ambient atmosphere.
C(Me)COO]2Sn (1) decomposes in air to spherical SnO particles of size 10–500 nm. Spin coating of 1 on Si or a glass substrate followed by heating at 200 °C results in a uniform film of SnO. The band gap of the produced SnO film and nanomaterial was determined by diffuse reflectance spectroscopy to be in the range of 3.0–3.3 eV. X-ray photoelectron spectroscopy indicates surface oxidation of the SnO film to SnO2 in ambient atmosphere.
- This article is part of the themed collection: In memory of Professor Kenneth Wade

 Please wait while we load your content...
Please wait while we load your content...